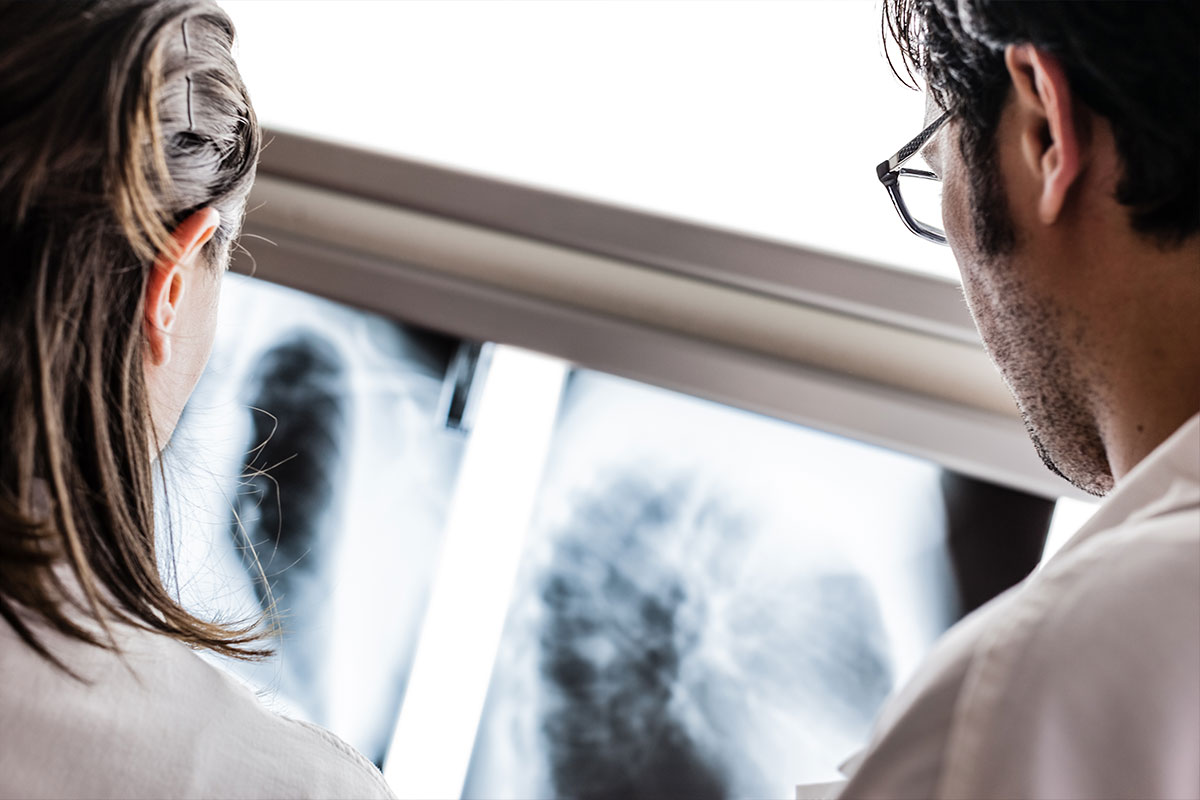
New studies indicate that the drug Elmiron, a medication already linked to health issues, may also cause vision problems in women with long-term use. The medication is often prescribed as treatment for urinary tract infections as well as osteoarthritis and other conditions.
Studies on Eye Damage
Several studies have been done on the side effects of Elmiron with more investigations underway. Information links Elmiron and vision damage. The amount of damage correlates with the quantity of the medication taken.
Elmiron was approved by the US Food and Drug Administration (FDA) to treat interstitial cystitis, which is a chronic condition of the bladder. It can cause pressure in the bladder along with pain. With long-term use, research indicates that it may be possible for Elmiron to cause pigmentary maculopathy.
This condition impacts the part of the retina known as the macula, which is the central part of the eye. It can cause issues with changes to light. The job of the macula is to protect the retina from UV light, which allows the person to see details better.
The Emory Eye Center, based in Atlanta, GA, made a report that showed that patients who had been taking the drug for about 15 years had changes in their macula. Another 10 patients had a diagnosis of interstitial cystitis and had been prescribed Elmiron. They also showed symptoms that corresponded with pigmentary maculopathy.
Another study was done by Kaiser Permanente which showed that about 24 percent of patients who had been prescribed Elmiron for a minimum of five years had damage to the eye.
Signs of Damage to the Eye
Symptoms associated with this condition include the following:
- Vision loss
- Difficulty with reading
- Vision that is distorted
While the condition can be preventable, it may not be treatable if it develops. This condition is just the latest to be related to Elmiron. Other conditions include the following:
- Amblyopia
- Optic neuritis and
- Retinal hemorrhage
Even after the patient discontinues the mediation, damage may occur. They may have difficulty adapting to an environment with low lighting. The condition can mimic macular degeneration in its later stages.
Anyone experiencing these symptoms after taking Elmiron should talk to their doctor about treatment. While it is never advised to stop taking prescription medication without consulting with their doctor first, it is important to arrange an appointment to discuss their symptoms with a medical professional right away. If they have taken Elmiron in the past and are experiencing vision changes, they will want to present that information to their doctor.
As the only FDA-approved treatment for IC, it must be continually taken, which means that thousands of patients may have been using Elmiron for years or even decades without knowing about the potential for vision damage. Even with the studies being conducted, Janssen Pharmaceuticals failed to warn the public until regulatory agencies applied pressure. Even when warnings were required in Europe and Canada, no changes were made to the labels in the US until June 2020. It is expected that more people will come forward with evidence of vision damage as the public becomes aware of the situation.